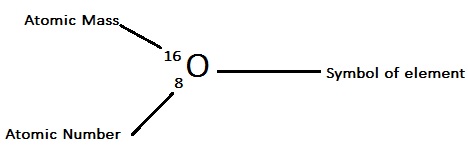
What is an atomic mass unit equal to? | Socraticġ atomic mass unit (amu) is the mass of a proton or neutron which is equal to 1.6726219 ×10−27kg. READ Can you start a column on the second floor? 1 amu is equal to a proton mass? 1 amu = Average of rest mass of protons and rest mass of neutrons. The mass of the atomic unit is symbolized as amu. What is the meaning of 1 amu? – Deep Advice Table of contents show What is 1 amu or 1u? Define one Atomic Mass Unit (amu) It is denoted by amu (atomic mass unit) or simply u. What is 1 amu equal to? Solved! - ScienceOxygenĪmu one = The aggregate of the rest masses of protons and neutrons. Hope this helps! For more explanation visit https///questions/13764/amu-and-g-mol-relation. So we just proved that an atomic mass unit is the same thing as grams per mole. Was this answer helpful? 0 0 Similar questions Amu one or and one is equal to Medium View solution > 1 atomic mass unit is equal to This question has multiple correct options Mediumġ2 amu/atom = 12 g/mol. Solution Verified by Toppr Correct choice is D) One amu is 121 of the mass of one carbon-12 atom. Therefore the mass of any isotope of any element is expressed in terms of the standard 12 C of AMU. 
This can be expressed as follows 1 AMU = 1.67377 x 10 -27 kilograms = 1.67377 x 10 -24 grams Carbon-12 is considered a reference for all atomic mass calculations. One AMU is the average of the rest mass of protons and the rest mass of neutrons. Suggest Corrections 16Ītomic mass unit (AMU or amu) - TechTarget 
The mass of one carbon atom is equal to the atomic mass unit (previously abbreviated as amu but now explained as u - unified mass) according to the latest IUPAC recommendation The mass of an atomic unit is represented with the symbol amu. What is an atomic mass unit equal to? - BYJUS This number is known as the atomic number which identifies the number of protons in the nucleus of ALL atoms in a given element. It is a unit of mass used to express atomic masses and molecular masses.Ģ.2 Atomic Number Mass Number and Atomic Mass Unitĭefine atomic mass unit Convert between amu and grams Atomic Number and Mass Number When you study the periodic table the first thing you might notice is the number on the symbol. In chemistry an atomic mass unit or AMU is a physical constant equal to one twelfth of the mass of an unbound atom of carbon -12. Other atoms generally do not have round-numbered atomic masses for reasons that are slightly beyond the scope of this article.ĭefinition of Atomic Mass Unit (AMU) - ThoughtCo By definition a carbon atom with six neutrons carbon-12 has an atomic mass of 12 amu. The atomic mass of a single atom is simply its total mass and is typically expressed in atomic mass units or amu. An atomic mass unit is equal to 1 / 12 of the mass of one atom of carbon-12 the most abundant isotope of carbon or 1.660538921 × 10 -24 grams.Ītomic number atomic mass and isotopes - Khan Academy Atomic mass unit | Definition Description Uses and FactsĪtomic mass unit (AMU) also called dalton in physics and chemistry unit for the expression of masses of molecules atoms or subatomic particles.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
